|
The results showed that the accuracy was very high ( Lowe 1977). Lowe provided a sixth-degree polynomial formula for computing the saturation vapor pressure of water from −50° to 50☌ and evaluated it against the Goff’s equation. Tabata suggested a quadratic formula that yielded the saturation vapor pressure with a mean percentage error of 0.17% within the temperature range from 0° to 35☌ ( Tabata 1973). Richards derived a four-degree polynomial for the saturation vapor pressure of water within the temperature range from −50° to 140☌ ( Richards 1971). The percentage error vs the observations was found to be less than 0.5% over the temperature range of 0°–35☌ ( Langlois 1967). Langlois developed a fractional formula for computing the saturation vapor pressure of water and compared the results with the observed values in Byers (1959) and the formula in Berry et al. Bosen gave a formula for calculating the saturation vapor pressure of water with reasonable accuracy in the temperature range from −51.1° to 54.4☌ ( Bosen 1960). There has been much research on formulating the saturation vapor pressure. The Goff–Gratch, Hyland–Wexler, and Wagner–Pruss formulas are tedious and inconvenient for use in calculating the saturation vapor pressure. 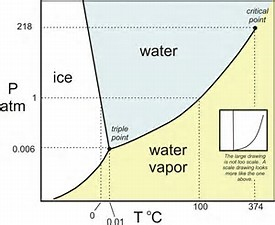
When large-scale computations are needed or temperatures with decimal points are reported, it is routine to calculate the saturation vapor pressure by using an accurate formula. The values of saturation vapor pressure can be obtained by looking them up in the reference tables. The IAPWS adopted it soon thereafter ( IAPWS 2011). developed a new equation, which has considerably less uncertainty, for calculating the saturation vapor pressure of ice ( Wagner et al. Wagner and Pruss developed a new formulation for the thermodynamic properties of ordinary water substance for general and scientific use, and later this formulation was adopted by the International Association for the Properties of Water and Steam (IAPWS) ( Wagner and Pruss 1993, 2002 IAPWS 2016). Later, this formula and the computed values were incorporated in the ASHRAE Handbook-Fundamentals ( Wexler 1976 Hyland and Wexler 1983 ASHRAE is derived from the American Society of Heating, Refrigerating, and Air-Conditioning Engineers). Wexler and Hyland obtained new laboratory data on the saturation vapor pressure of water and derived another lengthy formula. In 1966 the World Meteorological Organization (WMO) recommended its use, and the WMO meteorological tables were based on these formulas ( Letestu 1966). Goff and Gratch developed an accurate formula for calculating the saturation vapor pressure ( Goff and Gratch 1945), and modified this formula later ( Goff 1957). The saturation water vapor pressure, which is a function of air temperature, provides a basis for determining other thermodynamic properties of moist air (humidity ratio, specific enthalpy, specific entropy, specific volume, etc.). The latter condition is called saturation, a state of neutral equilibrium between moist air and the condensed water phase ( ASHRAE 2013, chapter 8). The amount of water vapor in moist air changes from zero to a maximum that relies on the temperature and pressure of atmospheric air. Moist air is a mixture of dry air and water vapor. The composition of dry air is relatively unvarying. 
Dry air exists when all water vapor has been removed from atmospheric air. Therefore, this new formula has significant advantages over the improved Magnus formula and can be used to calculate the saturation vapor pressure of water and of ice in a wide variety of disciplines.Ītmospheric air consists of a number of gaseous components (e.g., nitrogen, oxygen, carbon dioxide, inert gas, and water vapor). 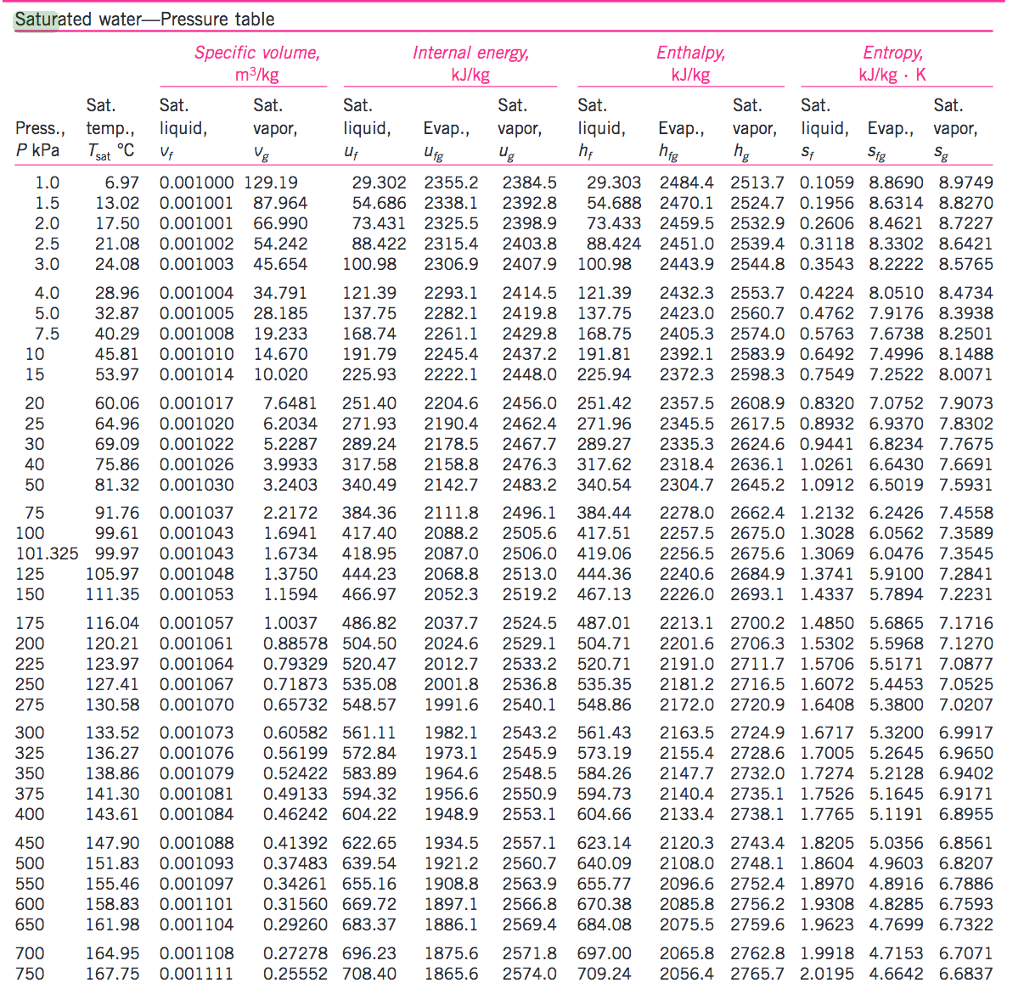
In addition, this new formula yields a mean relative error of 0.0005% within the commonly occurring temperature range (10°–40☌). In comparison with the International Association for the Properties of Water and Steam reference dataset, the mean relative errors from this new formula are only 0.001% and 0.006% for the saturation vapor pressure of water and of ice, respectively, within a wide range of temperatures from −100° to 100☌. This new formula is simple and easy to remember. In this study, a new formula has been developed by integrating the Clausius–Clapeyron equation. These formulas either are tedious or are not very accurate. A number of formulas are available for this calculation. It is necessary to calculate the saturation vapor pressure of water and of ice for some purposes in many disciplines.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
